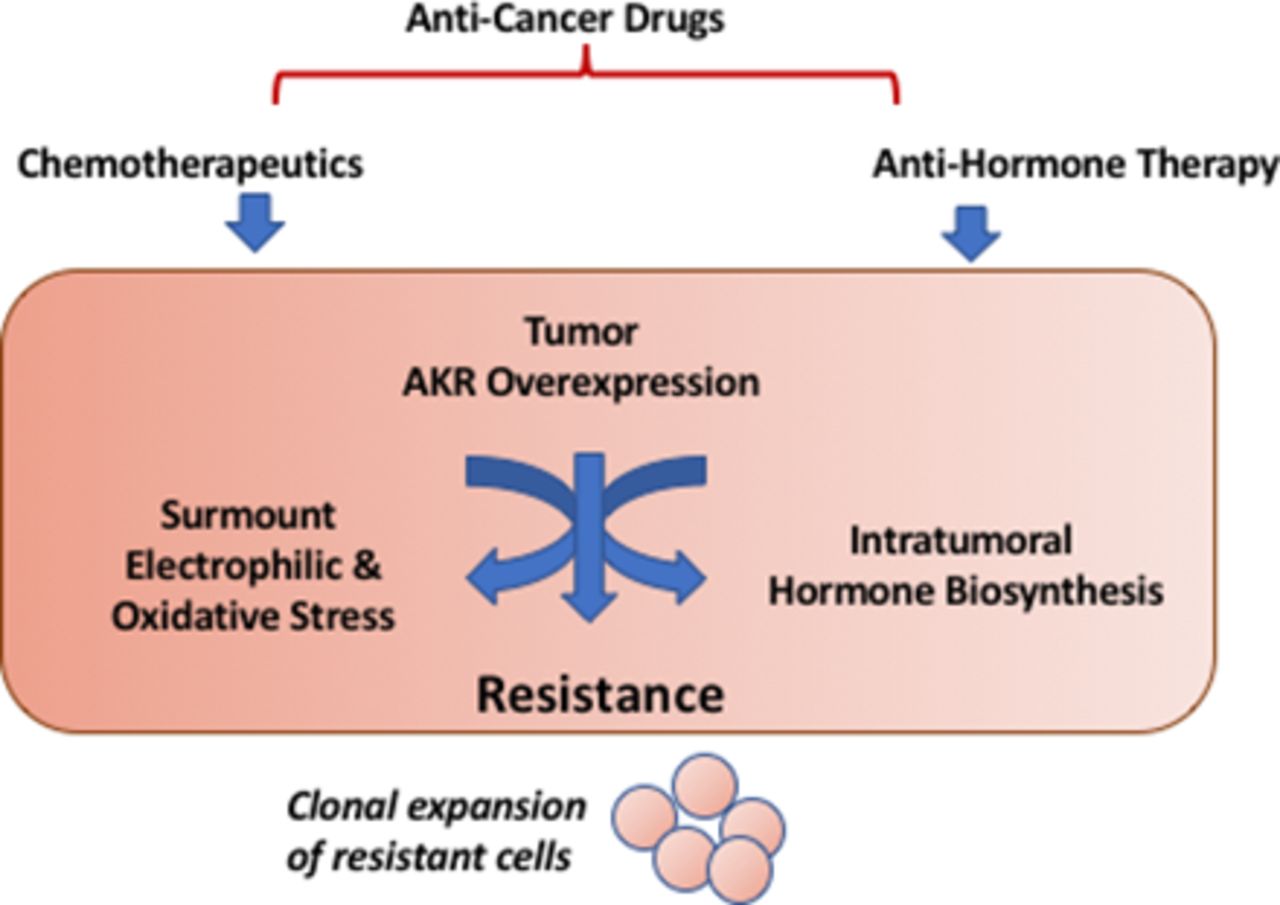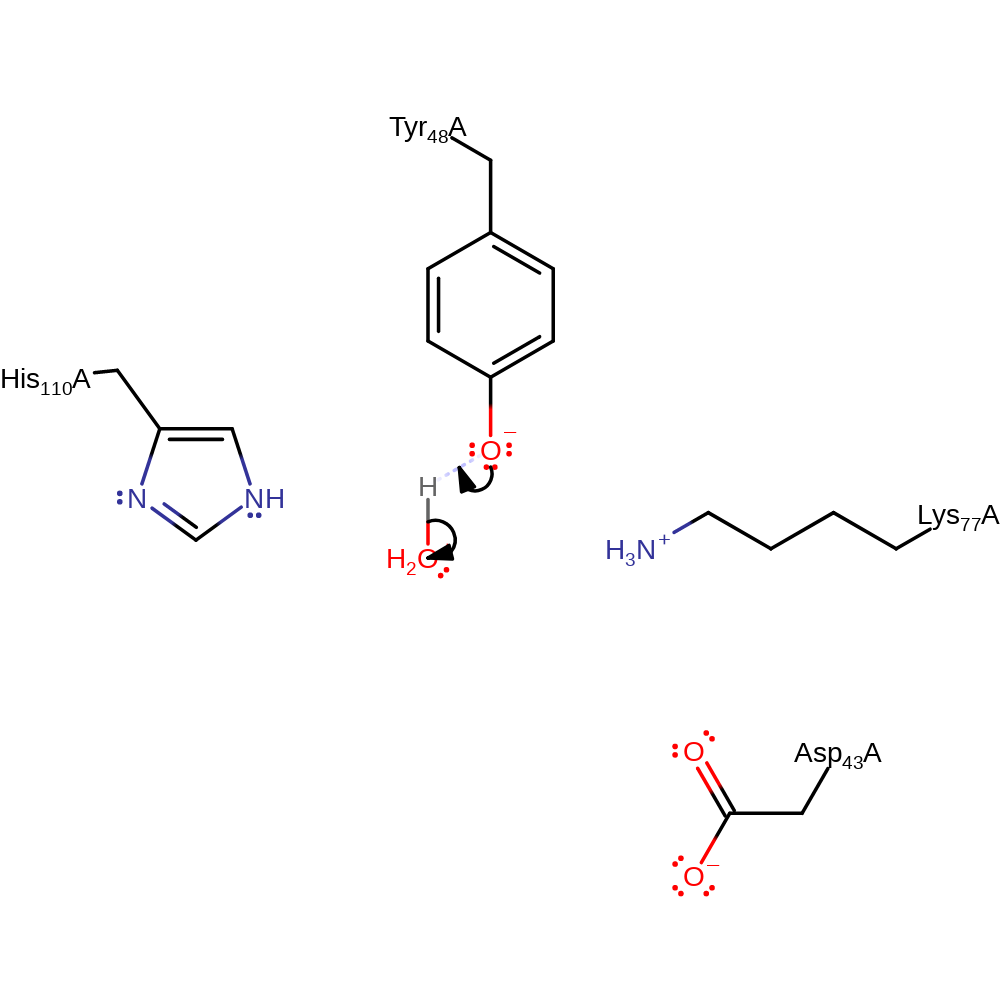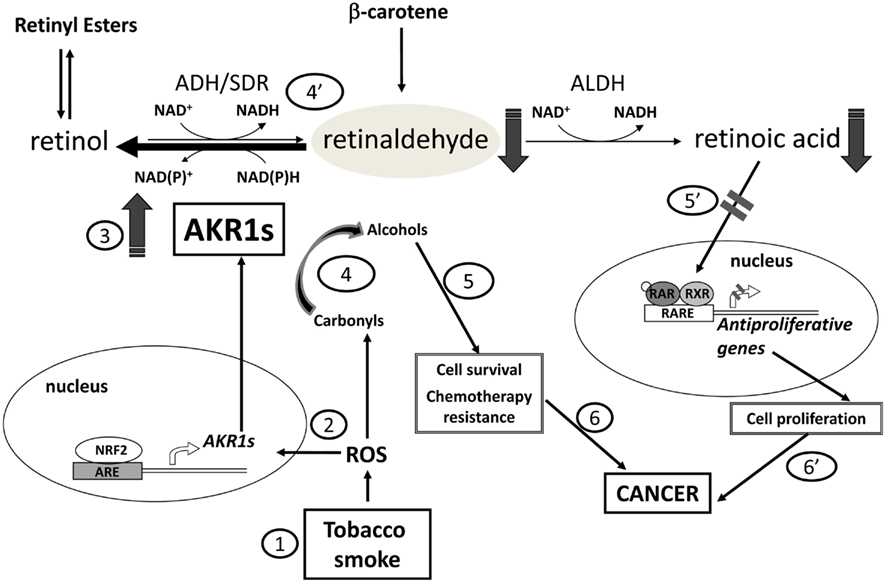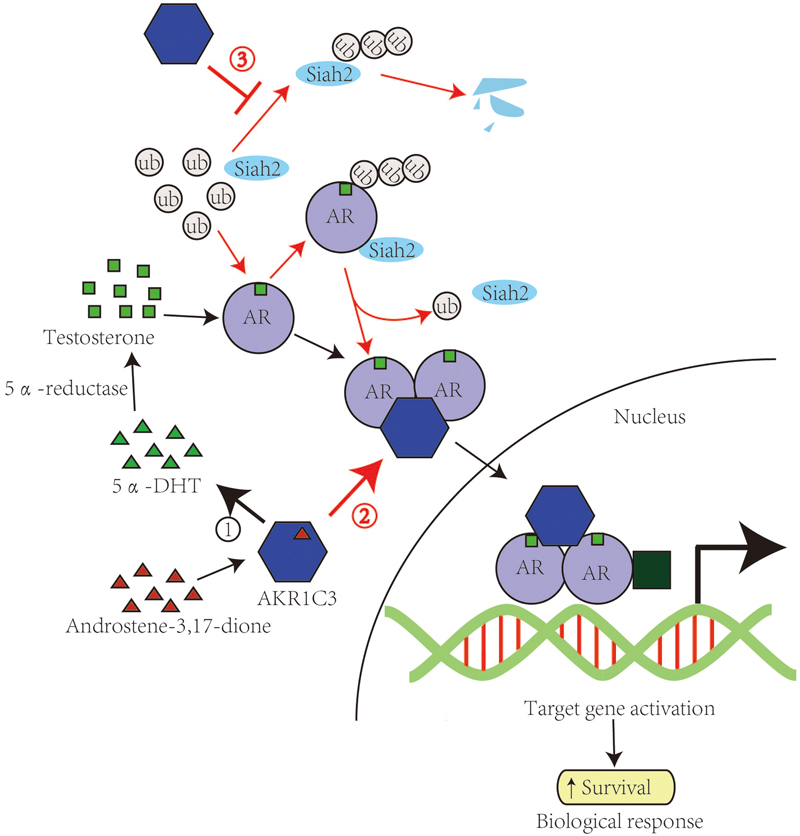
Frontiers | Aldo–Keto Reductase AKR1C1–AKR1C4: Functions, Regulation, and Intervention for Anti-cancer Therapy

Aldo-keto reductase 1B10 promotes development of cisplatin resistance in gastrointestinal cancer cells through down-regulating peroxisome proliferator-activated receptor-γ-dependent mechanism - ScienceDirect
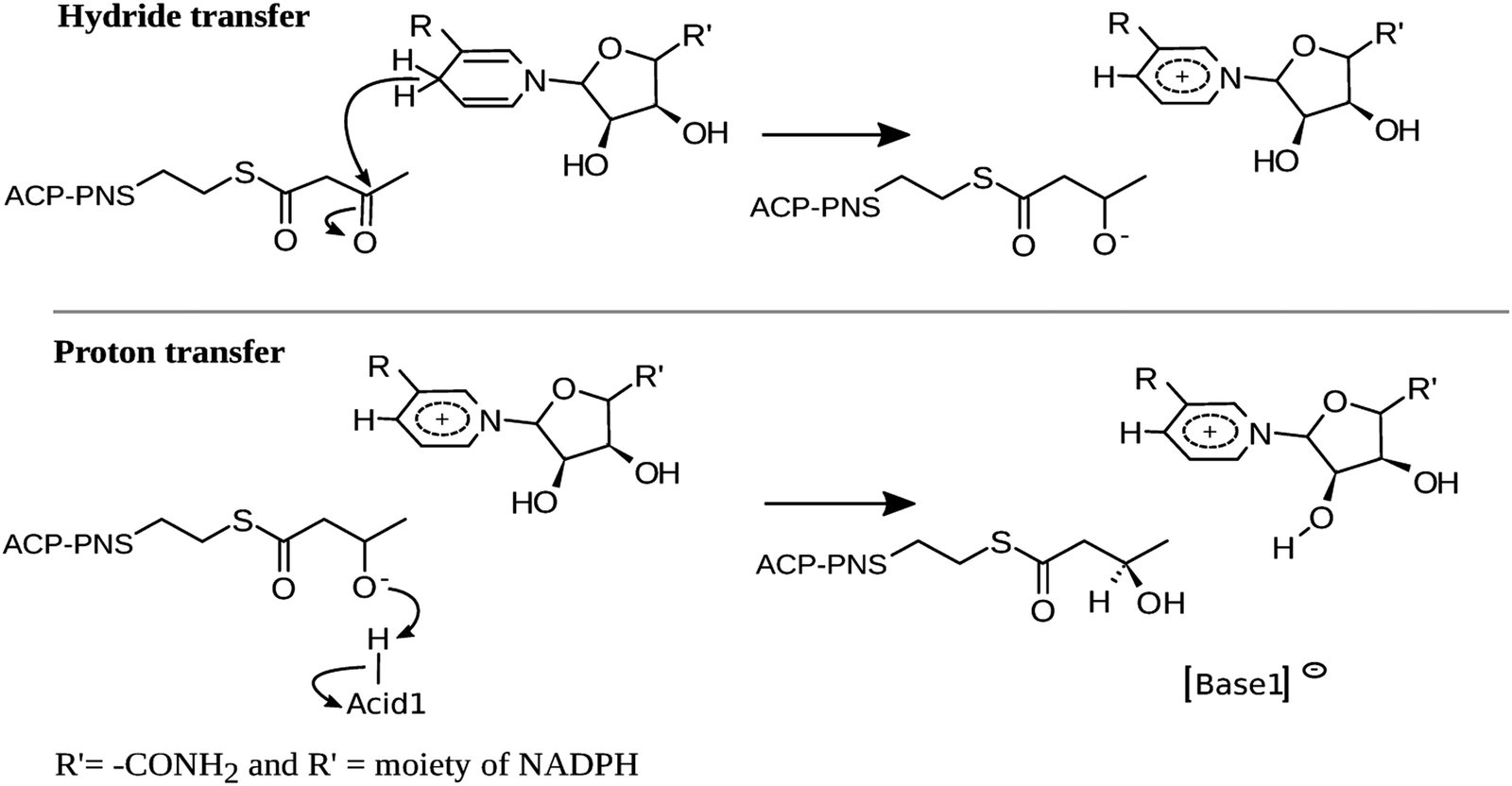
A QM/MM study of the reaction mechanism of human β-ketoacyl reductase - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C6CP07014K

Known ketoreductase catalyzed reactions for T-domain bound substrates... | Download Scientific Diagram

Evidence for the aldo-keto reductase pathway of polycyclic aromatic trans-dihydrodiol activation in human lung A549 cells | PNAS

Novel Aldo-Keto Reductases for the Biocatalytic Conversion of 3-Hydroxybutanal to 1,3-Butanediol: Structural and Biochemical Studies | Applied and Environmental Microbiology

Enantioselectivity and Enzyme-Substrate Docking Studies of a Ketoreductase from Sporobolomyces salmonicolor (SSCR) and Saccharomyces cerevisiae (YOL151w)

Identification and Characterization of a Selective Human Carbonyl Reductase 1 Substrate | Drug Metabolism & Disposition

Prostaglandin (PG) biosynthesis. Members of the aldo-keto reductase... | Download Scientific Diagram
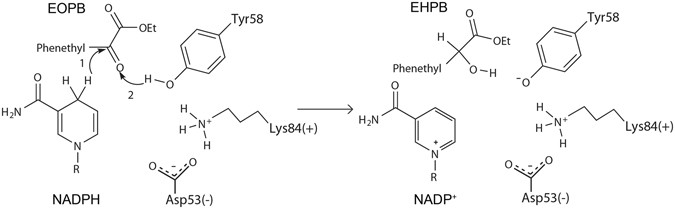
Semi-rational engineering of a thermostable aldo–keto reductase from Thermotoga maritima for synthesis of enantiopure ethyl-2-hydroxy-4-phenylbutyrate (EHPB) | Scientific Reports
Catalytic mechanism and substrate selectivity of aldoâ•'keto reductases: Insights from structureâ•'function studies of